Gene Maddaus Senior Media Writer Gov. Gavin Newsom has vetoed a bill giving unemployment benefits to striking workers, which was backed by the Writers Guild of America and SAG-AFTRA. “Now is not the time to increase costs or incur this sizable debt,” he wrote in his veto message Saturday.
New Fall Covid Vaccine Approved By FDA; Vaccinations Could Begin As Soon As This Week
11.09.2023 - 19:43 / deadline.com
The FDA today approved the new, fall Covid vaccine targeting omicron descendant XBB.1.5, which in over the summer was the most prevalent strain in the U.S. Last year’s vaccines targeted the original strain and an earlier omicron version. According to the AP, the new shots could be available as soon as this week.
As Covid numbers inch up across the country, U.S. officials are racing to get the new vaccine approved and available. The final step, would be approval by the CDC, which is set to issue recommendations tomorrow.
One question that has arisen during the process is whether the new shots will protect against the strains that have eclipsed XBB.1.5 in the past month or so. EG.5 is chief among them, which CDC models estimate made up roughly 21% of all cases nationwide on September 2, the most recent week for which there is forecasting. As of that date, XBB.1.5 had quickly shrunk to 3% of all variants. In thee western region comprised of California, Nevada and Arizona, XBB.1.5 made up more than 28% of all cases. See national chart below.
The good news is, per AP, that testing suggests the updated vaccine will offer protection good protection against EG.5, since it is genetically similar to a variant used to develop the vaccine.
“It doesn’t look like something that’s vastly different from what’s already been circulating in the U.S. for the past three to four months,” Andrew Pekosz, a professor of molecular microbiology and immunology at Johns Hopkins University Bloomberg School of Public Health told the New York Times last week.
Of more concern is a variant known as BA.2.86. While only a handful of cases have been identified in the U.S., scientists worry about the possibility of rapid spread because it is as heavily

Taylor Swift Scores For NFL & NBC In New ‘Sunday Night Football’ Promo; Superstar Expected In Attendance At NYC Game This Weekend
Having conquered the music biz, about to conquer the movie biz and proving one of this era’s most powerful economic and cultural forces, Taylor Swift is lending some of her superstar power to the NFL and NBC.

Jimmy Fallon, Seth Meyers Reveal That Lorne Michaels Helped Them Get Their Talk Shows
NBC didn’t want Jimmy Fallon to host Late Night at first.

Jimmy Fallon & Seth Meyers Weren't Even On NBC's Radar To Take Over As Hosts of 'Tonight Show' & 'Late Night'
Jimmy Fallon and Seth Meyers are recalling just how they got into the late night hosting game.

New York Film Festival Preview: 19 Films To Watch At 2023’s Fall Fest
There’s nothing like New York in the Fall— crisper air, pumpkin spice lattes, Cityside Pumpkin patch, the leaves changing in Central Park, and the New York Film Festival. What could be better? The 61st annual New York Film Festival kicks off in a few days, and there is a lot to look forward to.

New York Film Festival Preview: 19 Films To Watch At 2023’s Fall Fest
There’s nothing like New York in the Fall— crisper air, pumpkin spice lattes, Cityside Pumpkin patch, the leaves changing in Central Park, and the New York Film Festival. What could be better? The 61st annual New York Film Festival kicks off in a few days, and there is a lot to look forward to.

Watch Suicidal Tendencies’ Mike Muir fall off stage while singing ‘You Can’t Bring Me Down’
Suicidal Tendencies frontman Mike Muir fell off stage during the band’s show in Ontario, California on Saturday (September 23) – watch the video below.The hardcore punk legends were opening for US metal band Danzig when Muir took a tumble off the front of the stage at the Toyota Arena.The band were playing their opening song of the set, ‘You Can’t Bring Me Down’, which is taken from their 1990 album ‘Lights… Camera… Revolution!’.Footage captured by a fan shows the moment that Muir missteps as he walks near the edge of the stage. A few seconds later, with the aid of a technician, Muir is able to get back to his feet to complete the song, seemingly unaffected by the fall.As well as Danzig, the bands Behemoth, Twin Temple and Midnight were also on the bill.Next month, Suicidal Tendencies will play a number of shows in the US to celebrate the 40th anniversary of their debut self-titled album, including at New York City’s Terminal 5 on October 15 and two dates at The UC Theatre in Berkeley, California on October 19 and 20.

Late Night Talk Shows Could Return As Soon As Early October, Post-WGA Strike
Michael Schneider Variety Editor at Large As the WGA strike draws to a close, first stop on the road to normalcy will be late night — where producers are already plotting a return to air within the next two to three weeks. “We want to come back ASAP,” said one late night insider.

Rupert Murdoch Still Looking To Exert Political And Media Influence Despite Retiring As Fox & News Corp Chairman
The news that Rupert Murdoch was stepping down as chairman of Fox Corp. and News Corp. quickly triggered talk of his legacy, overshadowing one aspect of his announcement: Whether it comes to politics or to his companies’ media properties, he’s not going away.

Rupert Murdoch, The Creator Of Fox News, Is Stepping Down As Head Of News Corp. And Fox Corp.
Rupert Murdoch, the 92-year-old Australian media magnate whose creation of Fox News made him a force in American politics, is stepping down as leader of both Fox’s parent company and his News Corp. media holdings.

Pete Davidson adds 2 new NJ shows. Here’s how to get tickets now
Pete Davidson can’t get enough of the Garden State.After gigs in Atlantic City and Montclair earlier this year, the “King of Staten Island” just added two more NJ shows to his tour schedule later this year.First up, the “Saturday Night Live” alum is slated to headline at Red Bank’s Hackensack Meridian Health Theatre on Saturday, Nov. 18.After that, Davidson will take the stage the following night at Englewood’s Bergen Performing Arts Center on Sunday, Nov.

Gavin Newsom Says He Has Talked With WGA & Studios In Past Week & Will Meet Again With Them “Later This Week” – Watch
Gavin Newsom said in an interview today that he has been “deeply involved with talking” to both sides of the long-running Writers Guild strike and “we’re going to be meeting again later this week.”

California Senate Approves Unemployment Pay for Striking Workers
Gene Maddaus Senior Media Writer The California Senate voted on Thursday to grant unemployment benefits to workers who are on strike. The bill passed with a 27-12 vote. The Writers Guild of America and SAG-AFTRA have each expressed support for the bill.

California Senate Passes Bill To Give Striking Workers Access To Unemployment Insurance; Now It’s Up To Gov. Newsom
A bill that would make striking workers in California eligible to receive up to $450 a week in unemployment insurance benefits passed in the state Senate on Thursday by a vote of 27-12. Senate Bill 799, which passed in the state Assembly last month, now heads to the desk of Gov. Gavin Newsom, who can either sign it into law or veto it.
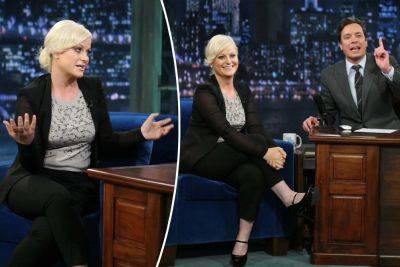
Jimmy Fallon’s heated exchange with Amy Poehler resurfaces amid toxic workplace claims
posted to X ( formerly Twitter), was initially covered in Tina Fey’s 2013 autobiography “Bossypants,” and depicted Fallon, 48, yelling at Poehler, 51, during a table read for the NBC variety show. “Amy Poehler was new to SNL and we were all crowded into the seventeenth-floor writers’ room, waiting for the Wednesday night read-through to start,” wrote Fey. “Amy was in the middle of some such nonsense with Seth Meyers across the table, and she did something vulgar as a joke.

Ezra Knight Reelected President Of SAG-AFTRA’s New York Local In Clean Sweep For Ruling USAN Party
Ezra Knight has been reelected president of SAG-AFTRA’s New York Local in a landslide victory that also gave his running mates a clean sweep of the election. Knight received nearly 73% of the votes cast in a four-person race for president. His four vice presidential running mates – Linda Powell, Anthony Rapp, Jim Kerr and Liz Zazzi – were also elected.

The Rolling Stones explain meaning of new ‘Hackney Diamonds’ album name
The Rolling Stones have explained the meaning behind the title of their long-awaited new album ‘Hackney Diamonds’. Watch the full press conference above.The legendary band announced their 24th studio record yesterday (September 6) and shared its lead single, ‘Angry’.

Huntington Beach Officials Working To Ban Mask And Covid Vaccine Mandates
A divided Huntington Beach City Council voted Wednesday to seek a ban on universal mask and Covid vaccine mandates in the city.

‘Anatomy of a Fall’ and ‘The Taste of Things’ Put Neon and IFC Back in Oscar Hunt with French Twist
Clayton Davis Senior Awards Editor Indie studios IFC Films and Neon are facing off with the hopes of one of their films being selected as France’s official submission to the Oscars for the international feature film prize. Neon aims to position Palme d’Or winner “Anatomy of a Fall” as the best option for the country. IFC is making its case for “The Taste of Things” from French-Vietnamese filmmaker Trần Anh Hùn, who won the director prize at Cannes.

Foo Fighters are headlining 5 festivals this fall. We found tickets
Foo Fighters will be performing.Dave Grohl’s hard-rocking group is headlining at five major music festivals from Sept. 1 through Oct. 18.Currently, they’re scheduled to close out Aspen, CO’s Jazz Aspen Snowmass on Sept.
